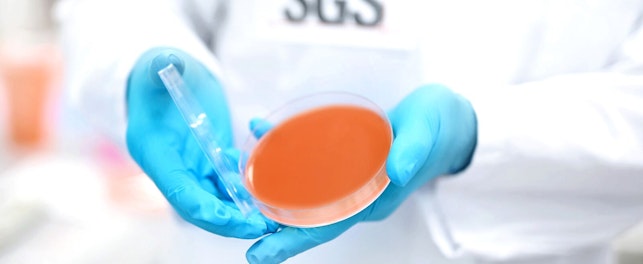
We're delighted to announce the launch of a dedicated Biosafety Level-2 microbiology lab within our testing facility in Phoenix, Arizona. Comprising around 4,000 square feet of the 12,000 square feet facility, the new lab will be focused on efficacy testing for hand sanitizers and antibacterial hand soaps.
This adds significant additional capacity to our testing capabilities, with studies expected to start in the new lab from June 2022 onwards.
As antimicrobial agents and compounds continue to serve an important role in protecting global health, we're committed to supporting our customers as they develop new products for the hand sanitizer and antibacterial hand soaps market. By adding capacity in Phoenix, we are able to offer more localized specialist testing for companies operating in the region.
High-Volume Efficacy Testing in the New SGS Lab
Our new laboratory facilities will provide a wide range of services to our customers in the antibacterial hand soap and hand sanitizer industries. These will include:
- The ASTM E2755 test (most commonly used for hand sanitizers) determines the bacteria-eliminating effectiveness of hand rub formulations used by healthcare personnel
- The ASTM E1174 test (most commonly used for antibacterial hand soaps) evaluates the effectiveness of handwash formulations for use by healthcare personnel
- Additional testing services such as in vitro testing and other standardized methods
SGS's Industry Leading Team
Our Phoenix-based facility will be led by Cosmetics & Hygiene Laboratory Manager Andrea Waggoner. Andrea is a member of the PCPC Microbiology Committee, the HCPA Antimicrobial Subgroup, and the ASTM E35.15 Antimicrobial Agents Subcommittee. She co-authored a 2007 paper on the effectiveness of antimicrobial soap for reducing the transmission of bacteria to food. Our lab's Director of Operations, Nancy Rodgers, also co-authored a study comparing antimicrobial agents with regular soaps.
The team's extensive research expertise in antimicrobial agents and their uses will ensure that the new lab provides our customers with the highest-quality testing services when assessing the efficacy of their products.
Andrea Waggoner said: "With research into antimicrobial agents more important than ever, I'm thrilled to be taking charge of this new SGS microbiology laboratory to provide efficacy testing for hand sanitizers and antibacterial hand soaps. It feels great to be able to offer our customers high-quality testing for their products as we continue the fight against viruses and bacteria."
We're proud to be able to provide our customers with trustworthy and reliable efficacy testing services that help them address the needs of the global community. With antimicrobial agents at the forefront of our collective fight against diseases, we're pleased to be able to contribute to our customers' efforts, making sure they can get their products to market swiftly and efficiently.
To find out more about our antimicrobial products and other testing services, contact us today.
Andrea Waggoner
Cosmetics & Hygiene Laboratory Manager
t: +1 (480) 682-0323
Janet Johnston
Cosmetics & Hygiene Business Development Manager, Client Services
t: +1 (972) 900-1107
About SGS
We are SGS – the world’s leading testing, inspection and certification company. We are recognized as the global benchmark for quality and integrity. Our 96,000 employees operate a network of 2,600 offices and laboratories, working together to enable a better, safer and more interconnected world.