Many first generation biologic products have reached, or are about to reach, patent expiry. In fact, IMS Health estimates that US 64 billion in global biologics sales will be off-patent by 2015. With the universal need for more affordable medicines in both developed and developing economies, many countries have established legal and regulatory pathways that allow the manufacture of copies of a patent-expired biotherapeutic product. As a result, ‘biosimilars’ – legally approved versions of an existing branded biologic – present a significant opportunity.
Biosimilar products are granted marketing approval on the basis of biosimilar analysis – side-by-side comparability with the originator. This requires comprehensive data from structural analysis and pre-clinical and clinical studies, which demonstrate that the biosimilar is ‘highly similar’ to the originator drug.
So how can you ensure the development of biosimilars that meets the required regulatory criteria?
Establishing biosimilarity
The development pathway for a biosimilar is unlike that of a novel biotherapeutic. There is an increased requirement for physicochemical analysis and testing – initially of batches of the originator to determine the Quality Target Product Profile (QTPP), then subsequently of the biosimilar, and finally head-to-head comparisons of the two.
This enhanced analytical effort may be rewarded in the reduced requirement for clinical trials – generally a Phase II trial is not necessary. However, careful consideration must be given to the planning of each phase of development. Many regulatory authorities throughout the world refer to a “step-by-step” approach to establishing biosimilarity.
Our services cover every aspect of biosimilar development – from originator characterization and biosafety testing, to full biosimilarity comparability studies, including physicochemical and biological assays, and clinical trials.
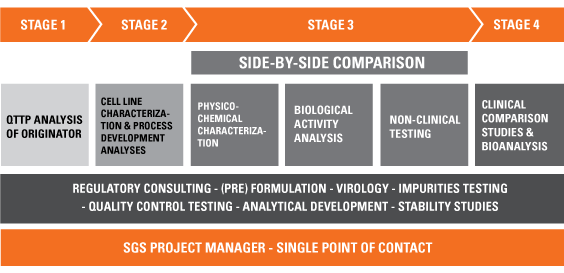
We can help you:
- Meet biosimilar regulations, such as the European Medicines Agency (EMA) guidelines on similar biological medicinal products and the U.S. abbreviated pathway under section 351(k) of the Public Health Service (PHS) act amended in the Biologics Price Competition and Innovation (BPCI) act
- Identify the primary and higher-order structure of proteins/glycoproteins, both originator and biosimilar at critical stages of development, helping you fulfill ICH Q6B and Q5E requirements
- Gain a range of clinical research services that can be trusted to deliver the highest standards throughout your clinical trials – from Phase I through Phase IV clinical trials to post-approval and the marketing of new products
Access world-leading biosimilar testing expertise
With extensive experience and expertise, we are a world leader in analytical, bioanalytical and clinical biosimilar testing, specializing in monoclonal antibody analysis. Our biosimilar services provide a dedicated point of contact and include regulatory advice and consultancy to aid you through the process.
Our wholly owned global network of 21 laboratories offers you centers of excellence in biopharmaceutical services and worldwide clinical research facilities. These include a clinical pharmacology unit and seven clinical research offices across the US and Europe.
As a result, we can perform the complete range of laboratory analysis and clinical trial studies, including comparative studies, to demonstrate the biosimilarity of your molecule.
To discuss how our biosimilar testing services can help you, contact us today.