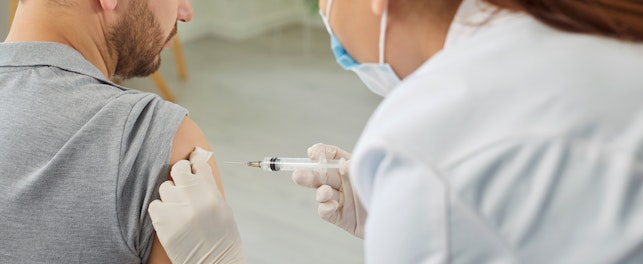
Participating in clinical research is a meaningful commitment. It requires motivation, availability, and a clear understanding of what is involved. ikbenpionier.be, our dedicated digital application, provides transparent and accurate information for healthy volunteers who are genuinely interested in taking part in clinical trials at our SGS Clinical Pharmacology Unit (CPU) in Edegem, Belgium.
Patients and healthy volunteers can engage in either Dutch or English, ensuring accessibility for a broad group of potential pioneers.

Why our pioneers choose to participate
Participants, or “pioneers”, as we call them, take part in clinical research for various reasons. Many are motivated by the ability to contribute to medical progress and support the development of new treatments. Others value the structured medical followup and close monitoring that are integral parts of a clinical study. Participation is also compensated, in line with applicable regulations and ethics approvals, acknowledging the required time and commitment.
Participation is always voluntary. Through ikbenpionier.be, candidates receive clear and structured information about the purpose of the research and the role pioneers play. This ensures that interest is based on understanding and realistic expectations, rather than curiosity alone.

Where clinical research meets everyday comfort
Clinical studies take place at our unit in Edegem in Belgium, located on the campus of University Hospital Antwerp (UZA). This location combines proximity to a major hospital environment with access to an established medical ecosystem, supporting participant safety, medical oversight and high-quality clinical execution.
The unit offers 110 beds in a modern, comfortable facility, equipped with all practical amenities volunteers may need during their stay. As studies are conducted onsite, pioneers must be able to travel to Edegem according to the study schedule. For longer inhouse stays, the CPU is designed to make time onsite as pleasant as possible. Where permitted by the protocol, participants can read, study or work remotely, allowing them to remain productive while contributing to research.
Throughout each study, participants are supported by experienced clinical teams who prioritize safety, comfort and strict adherence to international research standards.
A transparent and self-managed experience
The ikbenpionier.be platform is closely integrated with the Mijn Pionier app, which allows participants to manage their profiles, indicate availability and interact with studies in a structured, user-friendly way. This self-managed approach offers volunteers clarity and control while ensuring alignment with study requirements.
Before participation, candidates receive detailed information about procedures, study duration, potential procedural burdens and safety measures. This transparency supports informed decision-making and encourages sustained engagement throughout the study journey.
Supporting reliable recruitment through informed participation
Reliable recruitment begins with well-informed volunteers. When participants understand why they are taking part, what they can expect and where studies will be conducted, their engagement tends to be more consistent and predictable. ikbenpionier.be combines transparency, digital self-management and realistic expectations into one accessible platform. This benefits both pioneers and sponsors, ensuring that pioneers are prepared, motivated, and committed.
400 Broadacres Drive,
Suite 200, 2nd Floor,
Bloomfield, New Jersey, 07003,
United States